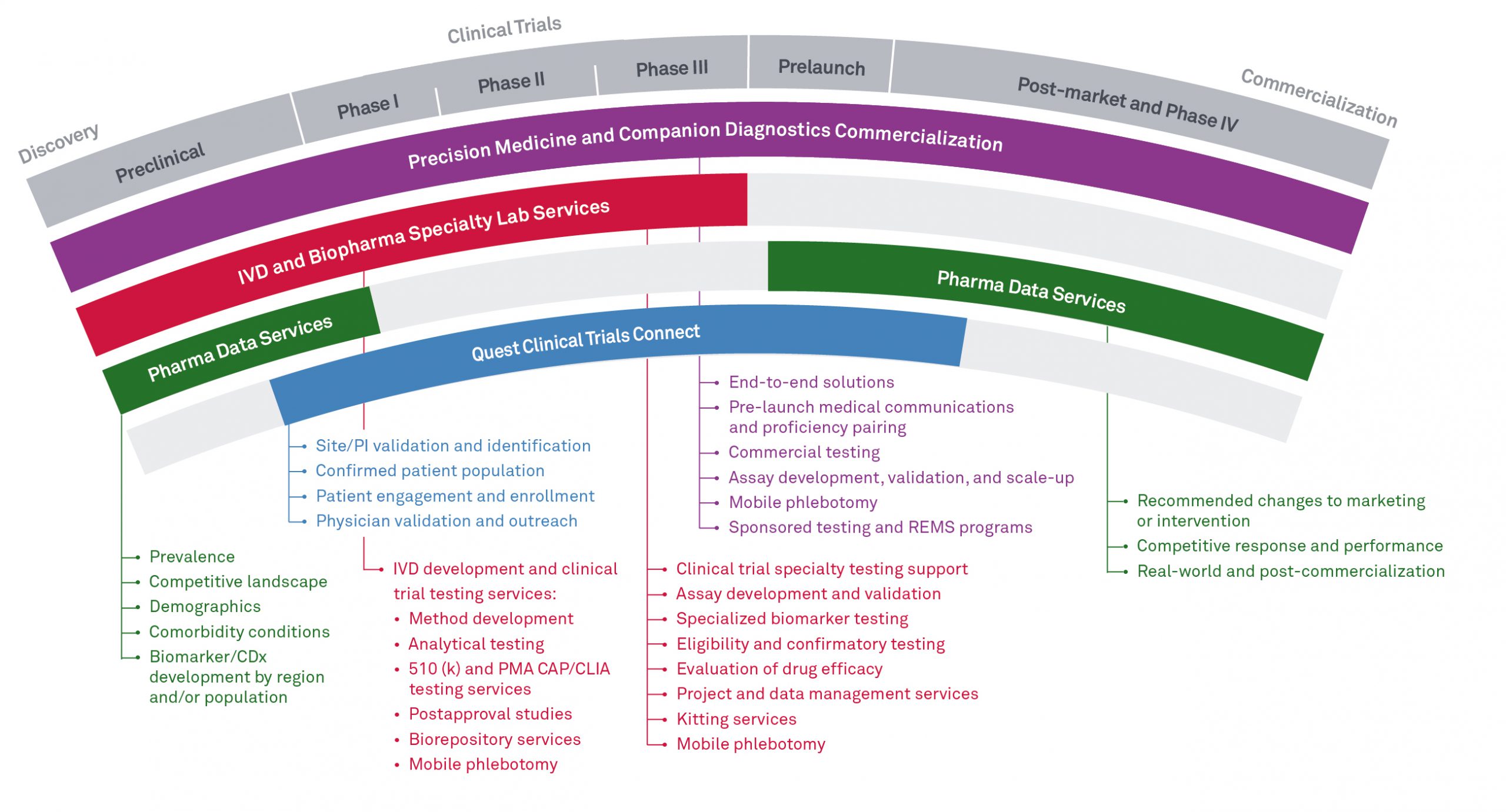
Where are you in the drug development continuum?
Quest Diagnostics Pharma Solutions supports you through all phases—discovery to clinical trials to commercial launch.

The power of the lab
Quest Diagnostics brings a depth of data, medical and scientific expertise, and analytics capabilities by centralizing resources that can:
- Enable the correlation between diagnostic insights with other data sets to understand prevalence
- Position your FDA submission for the best possible success through validation of emerging biomarkers
- Create necessary laboratory-developed tests to support your therapy
- Bring together physicians, patients, and Principal Investigators (PIs) to drive more effective and efficient clinical trials
- Drive clinical education and acceptance for a new product just reaching the market, and much more

Discovery
Choosing and validating the right idea to bring to market is key to long-term success.
Quest offers insights and services to help in the preclinical phase of development to identify and validate ideas for new therapies.
- The powerful combination of timely de-identified data, proven process, and seasoned expert analysis offered by our Pharma Data Services helps answer key questions about guidelines, disease management, prevalance, stratification, and disease profiles
- Our Precision Medicine team can give new insights into the latest in companion diagnostics, while our IVD solutions can help you prepare the idea for regulatory acceptance

Clinical trials
Finding a partner to make clinical trials more efficient and effective can cut down on the time it takes to commercialize a new therapy.
Quest offers support for all phases of clinical trials—from early-phase assay development to late-phase patient recruitment and site validation.
- Central lab services, end-to-end support, and customized testing solutions from our Companion Diagnostics team
- Efficient patient and physician recruitment and site validation are available through our healthcare analytics solution, Quest Clinical Trials Connect. Designed to grow with evolving demand, Clinical Trials Connect leads to a broad solution for cohort profiling, patient-initiated prescreening, and virtual trials

Commercialization
Creating awareness for your clinical therapies is only the first step to market acceptance.
Quest provides the right diagnostics to support new therapies and is key to helping generate sales.
- Our Companion Diagnostics experts work with you to deliver the diagnostics marketing and tests needed to drive acceptance, and to support the optimization of your commercial efforts
- Data Licensing and Analytics offers solutions to help you address health economics and outcomes research (HEOR) and real-world evidence and commercial needs. We help you understand how populations responds to intervention, growth trends, gap analysis, and marketing ROI analysis
Quest Pharma Solutions
Solutions for every stage of drug development
How we can help
Healthcare Analytics Solutions
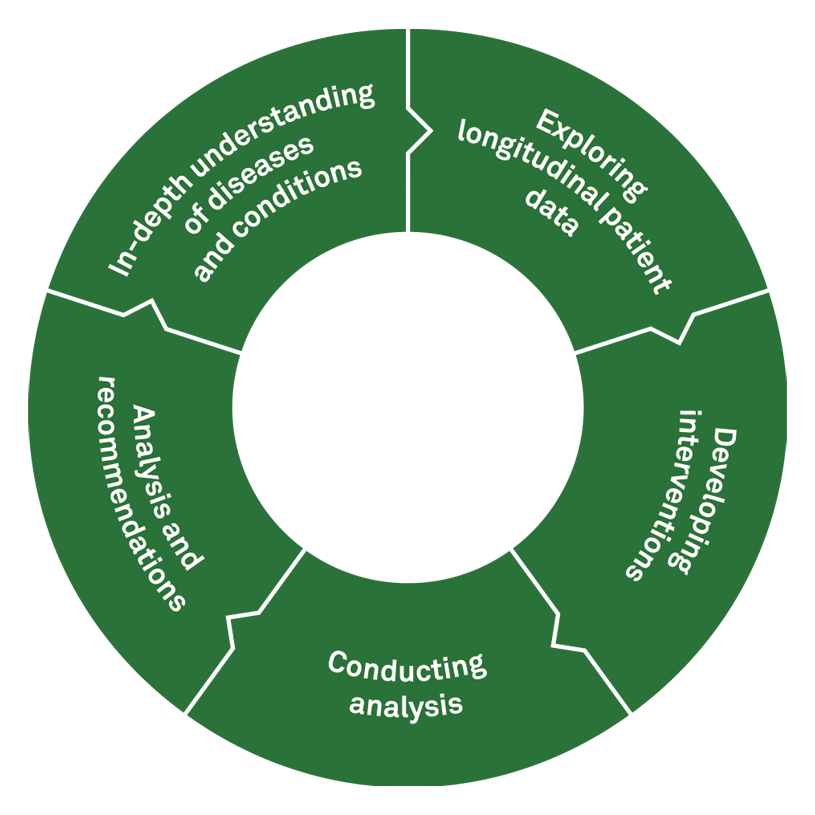
Quest Pharma Data Services
Delivering on the promise by simplifying complex data
Gain a competitive advantage with Quest Pharma Data Services. We combine our extensive inventory of laboratory, test, and patient data with client, third-party, and nationally relevant databases to create a holistic picture.a
Using Quest’s 60+ billion laboratory results, we apply an integrated approach to data analysis and reporting to help organizations devise a clear path forward:
- Identify the intervention opportunity
- Perform a detailed market assessment
- Monitor launch performance
- ROI analysis post-launch
- Recommended changes to marketing or intervention
a Only de-identified data is used in gathering insights into conditions and diseases
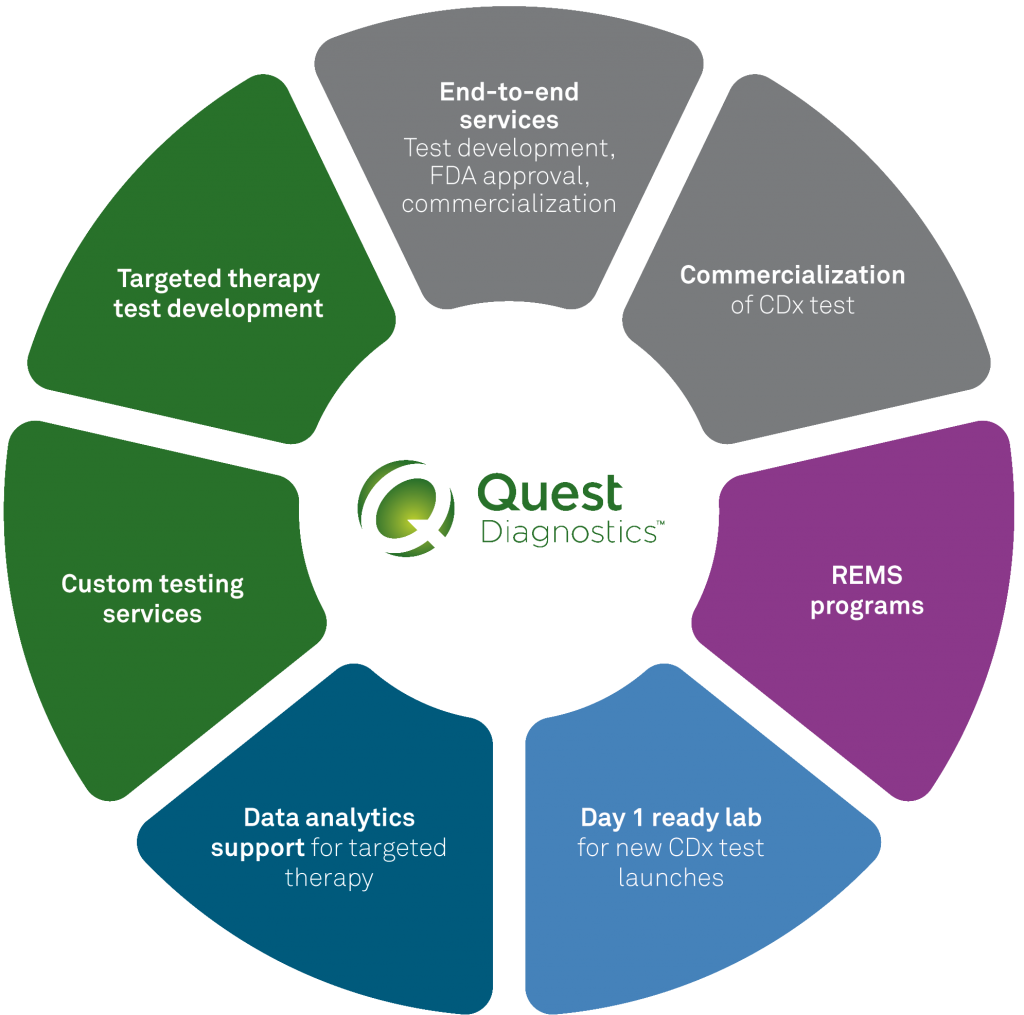
Tomorrow’s medicine today
Precision Medicine and Companion Diagnostics Commercialization Services
Quest Diagnostics Companion Diagnostics supports you through all phases of precision medicine, from assay development to commercial launch and post-launch analytic capabilities.
- End-to-end solutions
- Pre-launch medical communications and proficiency pairing
- Commercial testing
- Assay development, validation, and scale-up
- Mobile phlebotomy
- Sponsored testing and REMS programs

Supporting new therapies and emerging biomarkers
IVD Services and Biopharma Specialty Lab Services
Quest IVD and BioPharma Specialty Lab Services provide multi-site R&D and clinical testing services to support drug development and FDA clinical studies for IVD and BioPharma companies.
- Clinical trial testing for FDA, PMA and 510(k) submissions
- Post-approval studies
- Biorepository services
- Clinical trial wraparound services
- Assay development
- Validation testing
- Specialized biomarker testing
- Eligibility and confirmatory testing
- Evaluation of drug efficacy
Healthcare Analytics Solutions
Quest Clinical Trials Connect
Using insights to make clinical trials connections
Quest has relationships throughout healthcare—pharma, patients, physicians, health plans, and health systems—uniquely positioning us to make the connections needed to drive more effective clinical trials.
Efficient patient and physician recruitment and site validation are available today through Quest Clinical Trials Connect, and the solution is designed to grow with evolving demand, leading to a broad solution for cohort profiling, patient-initiated pre-screening, and virtual trials.
- Site/PI validation and identification
- Patient engagement and enrollment
- Physician validation and outreach


Testing solutions across the globe
Global Diagnostics Network
The Global Diagnostics Network (GDN) is a strategic working group of diagnostic laboratories, representing many of the world’s leading diagnostic companies.
Put Quest Pharma Solutions to work for you